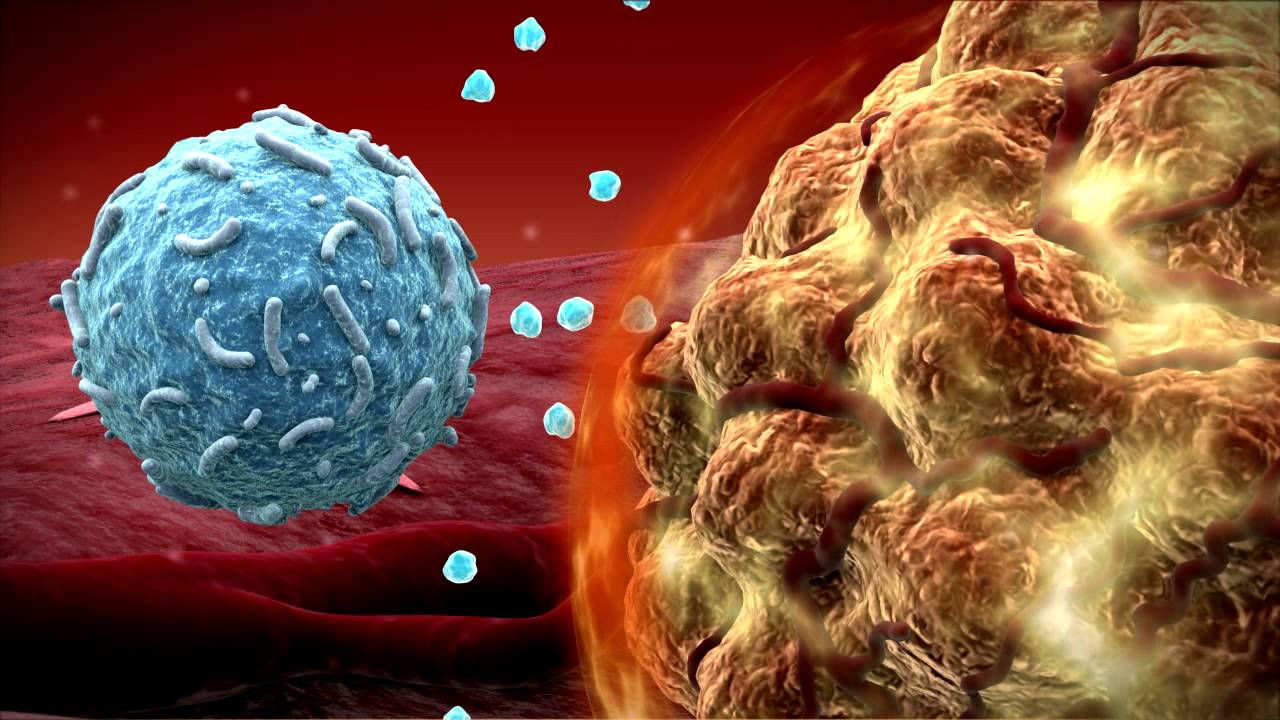
CAR T-cell therapies are one of the most promising and publicized immunotherapies, but limited in applicability. Two CAR T-cell immunotherapies approved by the FDA in 2017, Kymriah and Yescarta are limited to patients with specific types of blood cancer and failing prior conventional therapies. They are both designed for blocking a specific molecular target on cancer cells, either leukemia or lymphoma known as CD19. However, the CD19 molecule present on cancerous B cells is not the only one enabling them to multiply and grow. Rather, some patients with leukemia or lymphoma do not have CD19 on their cells, so the existing T-cell treatments do not work for them. Other patients, have CD19 at first and go into remission when treated, but then lose the protein and relapse within six months.
Researchers at the National Cancer Institute are engineering T cells to target the CD22 molecule (CD22-CAR) that is present on cancerous B cells, either in addition to CD19 or as the main driver of cancerous growth. This experimental therapy, if successful will provide an option to desperately ill leukemia patients who are relapsing on FDA approved CD19-CAR T-cell therapies. Additionally, it opens up the possibility of combination immunotherapy with two types of CAR T-cells administered simultaneously for enhanced results. These important developments are well covered in a New York Times Health article by author Denise Grady.
The Cancer Treatment Navigator team supports cancer patients in identifying the latest available treatment options and enabling access to targeted therapies and immunotherapies. Cancer patients often ask us about “Tumor melting T-cells”, described in press releases and news publications that highlight the breakthrough aspect, but often fail to explain the limitations of newly approved treatments. As scientific advisors to cancer patients, we explain nuances of approved immunotherapies and highlight experimental immunotherapies that might be more applicable to their specific cancer. Experimental immunotherapies require careful navigation given multiple clinical research trials running nationwide by hospitals and academic institutions. We encourage patients to leverage scientific research as part of their cancer care approach. Our team aims to emphasize balanced reviews and reports such as the New York Times article referenced above, so patients can be well educated and informed about cancer treatment choices.
Photo: Cancer fighting T-cell attacking Leukemia cell from New York Times; Eye of Science, via Science Source